In conversation with Praxis SCI Accelerate Cohort Member, Inteligex

Bioengineered Neural Stem Cell-based Products
Interview with Dr. Michael Fehlings, President and Founder
Inteligex is a Canadian regenerative medicine company developing stem cell treatments for SCI and other CNS diseases. Inteligex has developed two proprietary neural stem cell-based products (one targeting the acute stage of SCI and one focusing on the chronic stage). These cells are designed to integrate into the injury microenvironment at the specific spinal cord level. Inteligex’s cells have demonstrated significantly enhanced functional recovery in a high-fidelity preclinical model of SCI. Inteligex is now working to translate these human stem cell technologies for use in the clinic.
What led you (education, experience, people) to a career focused on medical technology and, in particular, medical technology for people living with SCI?
When I was a neurosurgical resident, SCI wasn’t really on my radar until I became involved with Dr. Charles Tator [whose lab was the first in Canada to study acute SCI from a basic science perspective].
Back then, when an individual sustained an injury there wasn’t really anything that could be done. I would be doing rounds in the clinic and see these young people who, just yesterday, were active, fit and doing everything they wanted to in their lives, and then the next day they’re laying there with a significant injury, a catastrophic impact on their life that isn’t reversible in any way.
Inteligex is a spin-off company resulting from my research over the last 20 years at UHN. The company was formed in 2018, with the goal of translating this research from bench to bedside.
What was your biggest learning/takeaway from the program?
My team and I spent a lot of time developing the focus group questions with the mentors – this process provided a lot of valuable feedback from their own experiences living with spinal cord injury and perceptions of cell therapies. It complemented what I see and hear from patients in the clinic, but it was nice to have the discussions in a different environment where everyone is focused on making Inteligex the best that it can be.
With cellular therapies, there are many products available for treating different conditions, and the coverage in mainstream media is often misleading. So, when we received feedback from the focus group, it was clear that each participant had a very different opinion, based on their life experiences and what they’ve been exposed to, about the potential utility of our product for SCI. The focus group highlighted our need to be very clear in our messaging. For example, we need to make sure that the pipeline for using these cells is well defined, as well as clearly communicating the research we have done that allowed us to refine our cells and prepare them for clinical use.
The focus group participants and mentors were very honest, so it was nice to have them playing the devil’s advocate in a way. It helped us to strengthen our messaging in a supportive environment and hopefully sets us up for future success.
Inteligex’s preclinical findings demonstrating improved forelimb strength, better dexterity, and reduced pain can bring life changing improvements if successfully translated to individuals living with spinal cord injuries.
Are there any shifts you plan on making in your company’s structure and/or business strategy in the next few months?
There was a lot of encouragement from the mentors for using Accelerate program funding to support knowledge translation initiatives. To this end, we’re creating a professional video discussing our technology and the critical need to bring products like ours to patients.
The program has encouraged us to invest more in patient education, to show the commercialization pathway, and also to highlight our unique selling points. We have spent years performing high-quality research to better understand the injury cascade and why other cell therapies have been unsuccessful. We have leveraged this information to create our product, which can be tailored to an individual’s specific level and stage of injury, yielding improved outcomes. This is the story we really want to share with our funders and end users.
Is there a story you’d like to share from the program when your engagement with a person with lived experience (clinician, person with SCI, care provider) was impactful to you or your company’s development?
In our discussions with the program mentors, they consistently highlighted the need for gains in independence for people living with SCI. Enhanced forearm or finger function could make a major difference in independence and quality of life, and this message underpins everything that we’re doing in the lab with respect to developing our therapy. In the mainstream media, finding a miracle cure to restore walking and all function is often portrayed to be the goal, but at this stage we are looking for a few incremental gains that, when summed, can dramatically alter a patient’s life.
When you look at data from medical research, you will often see results that can appear relatively minor to a lay person who has not spent much time in the clinic. And occasionally this creates the perception that a therapy doesn’t work or that research is moving too slow. But, in Inteligex’s case, our preclinical findings demonstrating improved forelimb strength, better dexterity, and reduced pain can be life changing improvements if successfully translated to individuals living with spinal cord injuries. That is the difference between someone requiring 24/7 care versus being able to feed themselves, dress themselves, and to be more comfortable.
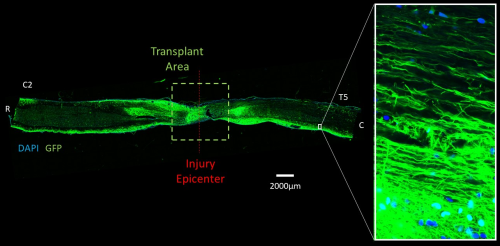
An example of how Inteligex’s cells work when they are transplanted into a preclinical model of spinal cord injury. Once transplanted into the injured spinal cord, Inteligex’s cells are able to extend long axons in the spinal cord and across the injury site.